Spinraza (nusinersen), a breakthrough treatment for spinal muscular atrophy (SMA), is transforming patient survival worldwide. But the cost of SMA treatment varies dramatically between countries, from heavily subsidised access in China and Australia to extremely high list prices in the United States.
Over the past decade, the treatment landscape for Spinal Muscular Atrophy (SMA) has undergone a profound transformation. Once considered one of the most devastating inherited diseases affecting children, SMA is now treatable through a new generation of genetic-targeted therapies that address the condition at its molecular source.
Yet while scientific progress has accelerated, access to these life-changing medicines varies dramatically depending on where patients live.
Across Asia, the rapid growth of advanced medical treatments is also reshaping regional healthcare systems and cross-border treatment options. Developments in Vietnam’s hospital sector and emerging treatment access are increasingly covered by specialised healthcare platforms such as Medical Tourism Vietnam
One drug in particular ” Nusinersen (Spinraza)” illustrates how national healthcare systems negotiate the cost of breakthrough treatments. A comparison between China, Australia, and the United States reveals three very different approaches to paying for life-saving innovation.
Understanding Spinal Muscular Atrophy
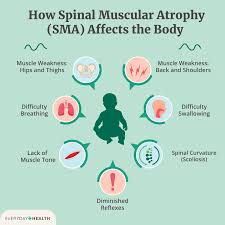
Spinal Muscular Atrophy is a rare inherited neurological disease that causes progressive muscle weakness and atrophy. It is one of the most common genetic causes of infant mortality worldwide.
The condition occurs when mutations in the SMN1 gene prevent the body from producing sufficient survival motor neuron (SMN) protein, which is essential for maintaining motor neurons in the spinal cord. As these neurons deteriorate, patients gradually lose the ability to control voluntary muscles such as those required for walking, swallowing, and breathing.
A related gene, SMN2, produces smaller amounts of the same protein. The number of SMN2 copies a person has can influence how severe the disease becomes.
SMA is inherited in an autosomal recessive pattern, meaning both parents must carry the defective gene for a child to develop the disorder. When both parents are carriers, each pregnancy carries a 25 percent chance of producing an affected child.
Doctors typically classify SMA into four types:
| Type | Age of Onset | Key Characteristics |
|---|---|---|
| Type 1 | Birth–6 months | Most severe form; infants cannot sit and develop severe breathing difficulties |
| Type 2 | 6–18 months | Children can sit but cannot walk independently |
| Type 3 | Childhood | Milder form; patients can walk but may lose mobility over time |
| Type 4 | Adulthood | Rare and slow-progressing muscle weakness |
For decades, treatment options were largely limited to supportive care. That began to change with the arrival of genetic-targeted therapies.
The Drug That Changed SMA Treatment
Spinraza became the first disease-modifying therapy approved for SMA.
Rather than replacing the faulty SMN1 gene, the drug works by modifying how the SMN2 gene is processed, enabling it to produce more functional SMN protein.
The therapy is delivered through intrathecal injection directly into the spinal canal, allowing the medication to reach motor neurons in the central nervous system.
Treatment begins with an intensive loading phase:
- Four injections within the first two months
- Followed by maintenance injections every four months for life
Clinically, Spinraza has dramatically improved outcomes for many patients. Financially, however, the therapy quickly became known as one of the most expensive drugs in modern medicine.
China: From ¥700,000 Injections to National Reimbursement
China offers perhaps the most dramatic example of how government negotiation can reshape access to expensive medicines.
When Spinraza first entered the Chinese market in 2019, it was priced at approximately ¥700,000 per injection. For most families, the drug was effectively unaffordable.
The turning point came during the National Healthcare Security Administration (NHSA) price negotiations in 2021.
Chinese media later described the negotiations as “soul bargaining”, reflecting the intensity of discussions between regulators and the pharmaceutical manufacturer.
The outcome was remarkable.
The price per injection fell to approximately ¥33,000, representing one of the steepest price reductions ever negotiated for a rare disease treatment.
Spinraza was then included in China’s National Reimbursement Drug List (NRDL), allowing the therapy to be partially covered under the national health insurance system.
Today, the approximate annual treatment cost is roughly:
¥132,000 (about USD $18,000)
For insured patients, the majority of that cost is reimbursed, leaving a comparatively small out-of-pocket payment.
For many Chinese families, a treatment that once seemed impossible has become accessible.
United States: High Prices and Insurance Dependence
The United States presents a markedly different model.
Spinraza received FDA approval in 2016 and is widely available across the country. However, the American healthcare system relies heavily on private insurance coverage.
The official list price remains extremely high:
- First year treatment: approximately $750,000 USD
- Subsequent years: roughly $375,000 annually
Patients rarely pay the full amount directly, but their out-of-pocket cost depends heavily on the structure of their insurance plan.
Some families pay relatively modest co-payments, while others with high-deductible policies may face significant cost-sharing. Patients without insurance face the full price.
Behind the scenes, insurers often negotiate confidential rebates with pharmaceutical manufacturers. Even so, the cost remains among the highest for any long-term therapy currently available.
Global Cost Comparison
| Country | System | Approximate First-Year Cost | Typical Patient Cost |
|---|---|---|---|
| China | National insurance (NRDL) | ~¥132,000 RMB (~$18,000 USD) | Small co-payment |
| Australia | PBS universal subsidy | ~$104,000 AUD per vial | $31.60 / $7.70 prescription |
| United States | Insurance-based system | ~$750,000 USD | Varies widely |
The Rise of Regional Medical Travel
Across Asia, the rapid expansion of advanced therapies is also beginning to shape a new form of regional medical travel.
As treatment costs diverge between countries, patients and families increasingly research options beyond their domestic healthcare systems. Nations across Southeast Asia are investing heavily in modern hospitals, specialist training, and international accreditation in order to attract cross-border healthcare demand.
Vietnam in particular has been steadily emerging as a regional healthcare hub, combining internationally trained medical specialists with comparatively lower treatment costs and expanding private hospital infrastructure.
For readers interested in developments in Vietnam’s healthcare sector — including hospital systems, medical innovation and emerging treatment access — further reporting is available through the Medical Tourism Vietnam platform.
The Global Debate Over the Cost of Innovation
Spinraza highlights a broader global challenge facing healthcare systems.
China relied on centralised price negotiations to force dramatic cost reductions. Australia subsidises the therapy through public healthcare funding. The United States depends largely on insurance negotiations within a market-driven system.
All three countries provide access to the same life-saving therapy — but the financial pathway to obtaining it is dramatically different.
As a new generation of gene therapies and precision medicines enters the market, some priced in the millions of dollars, governments and healthcare systems worldwide face an increasingly urgent question:
Who should ultimately bear the cost of medical breakthroughs?
Related Healthcare Developments in Asia:
Readers interested in developments in Asia’s healthcare sector may also find these reports useful:
• Vietnam approves advanced cancer immunotherapy expanding treatment access
• The rise of Vietnam as a regional medical treatment hub
• Healthcare innovation and hospital expansion across Southeast Asia
Data Sources
Pricing and regulatory information referenced in this article is based on publicly available data from national health authorities, pharmaceutical pricing reports, and regulatory agencies including the U.S. Food and Drug Administration (FDA), China’s National Healthcare Security Administration (NHSA).
Source
Further reporting on healthcare developments across Vietnam and Asia can be found at
Medical Tourism Vietnam
Information on treatment access and patient services in China is available through
GoBroad Healthcare Group
More healthcare reporting is available at Medical Tourism Vietnam.
Leave a comment